The final frontier to improving low-pain injections
25 January 2024
Most people dislike injections. In some cases, that aversion comes from the pain experienced upon injection, and this discomfort can have important consequences for the patient, the pharma company, and the device manufacturer. This article analyses the causes of injection pain, identifies an under-explored area for improvement, and presents a concept that might minimise discomfort.
Painful injections are a problem
The pain or discomfort of an injection can dissuade patients from getting essential medication, whether administered by a simple syringe and needle, prefilled syringe, on-body injector or autoinjector. This article will focus on autoinjectors as a concrete example, but the concept presented below could be applied to any injection system.
An autoinjector is a handheld device that allows its user to inject themselves with a drug more easily than with a simple syringe. Some autoinjectors are intended for emergency treatment, such as epinephrine administration against anaphylaxis; while others are used to treat chronic conditions, like beta interferon for multiple sclerosis. The global autoinjector market was valued at $1.5 billion in 2019 [1].
Despite the importance of autoinjectors to people’s lives and the commercial scale of the industry, there is still huge potential to improve the design and user experience of these devices. Making injections less painful is clearly a good thing in itself, and could also be expected to improve patient compliance.
Where can we most improve the autoinjector user experience?
To pinpoint the best strategic opportunities for improvement of an autoinjector’s user experience, we can use a user journey map. This is a visual way to break down the user steps and get an overview of the resulting user experience.
As an example, we created a user journey map for a multiple sclerosis patient self-administering the drug beta interferon using an autoinjector (Figure 1). Analysis of this map shows that these patients face two main difficulties (circled in green in the figure).
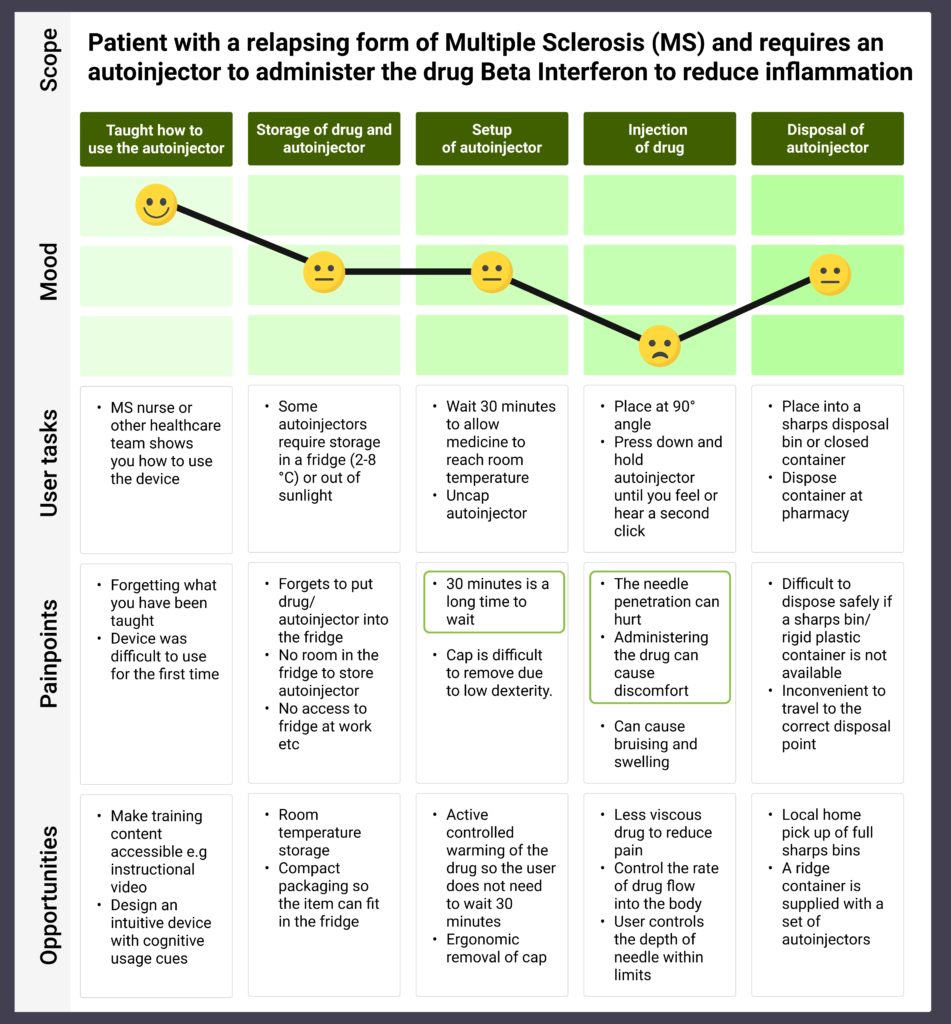
Figure 1: User journey map of a patient with a relapsing form of multiple sclerosis.
Firstly, several autoinjectors require refrigerated storage, such as the Avonex Pen which must be stored at 2–8°C [2]. Patients are therefore instructed to wait 30 minutes to allow the medicine to reach room temperature. This waiting step may be seen as inconvenient, and users might choose to skip it because they want to use the device immediately. However, skipping the step means the injected drug will be more viscous than intended and will cause the user additional pain.
Secondly, the penetration of the needle and dispensing of the drug are painful. This pain can be particularly difficult to endure from autoinjectors that are used multiple times a week, as is the case for Rebif beta interferons [3].
What makes injections painful?
Having identified these two strategic areas for strategic improvement, we can now analyse their root causes to understand how those improvements could be made.
Overall there are two sources of pain in an injection: the insertion of the needle, and the subsequent injection of the drug. The drug injection can cause pain from physical stress placed on surrounding tissue, the drug’s chemical properties or from a temperature difference between the drug and the tissue. Although there is scope for improvement in each of these areas, most of the associated design changes are limited by manufacturing techniques or hampered by constraints on drug formulation (Figure 2). Efforts are being made in all these areas, but the green path highlighted in the figure shows a relatively unexplored frontier for pain reduction: the temperature difference between the drug and the body. This led us at Springboard to create the concept described below.
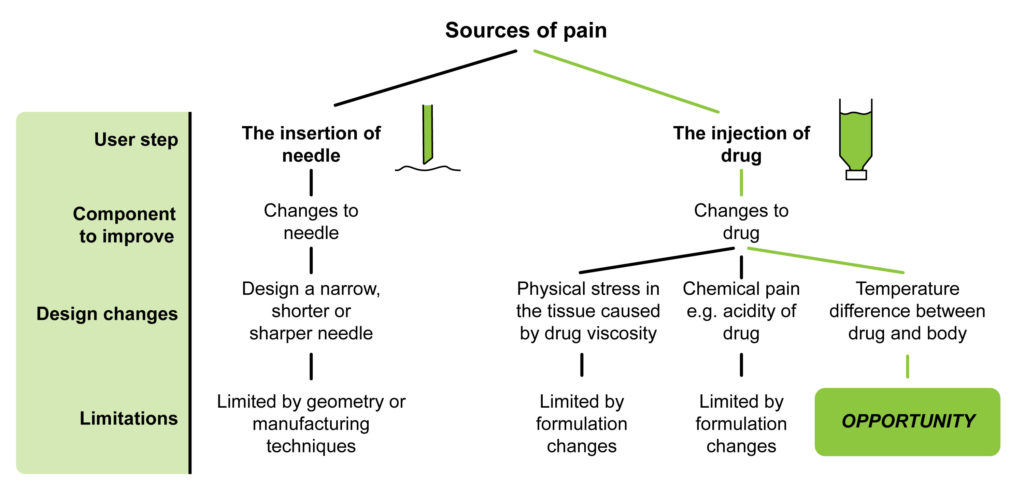
Figure 2: Sources of pain from insertion of needle and injection of drug suggest numerous avenues for improvement. Reducing the temperature difference is a relatively untapped area for improvement.
How could the drug approach body temperature before injection?
Our concept is essentially to actively warm the drug approximately to body temperature without the use of expensive and inconvenient powered devices. One possible implementation could employ iron filings in a similar way to common hand warmers: when exposed to oxygen the filings oxidise, and the energy released in this exothermic reaction could be used to heat the drug. Specifically, as shown in Figure 3 the device could be designed such that the released heat is transferred to the housed needle end of the syringe.
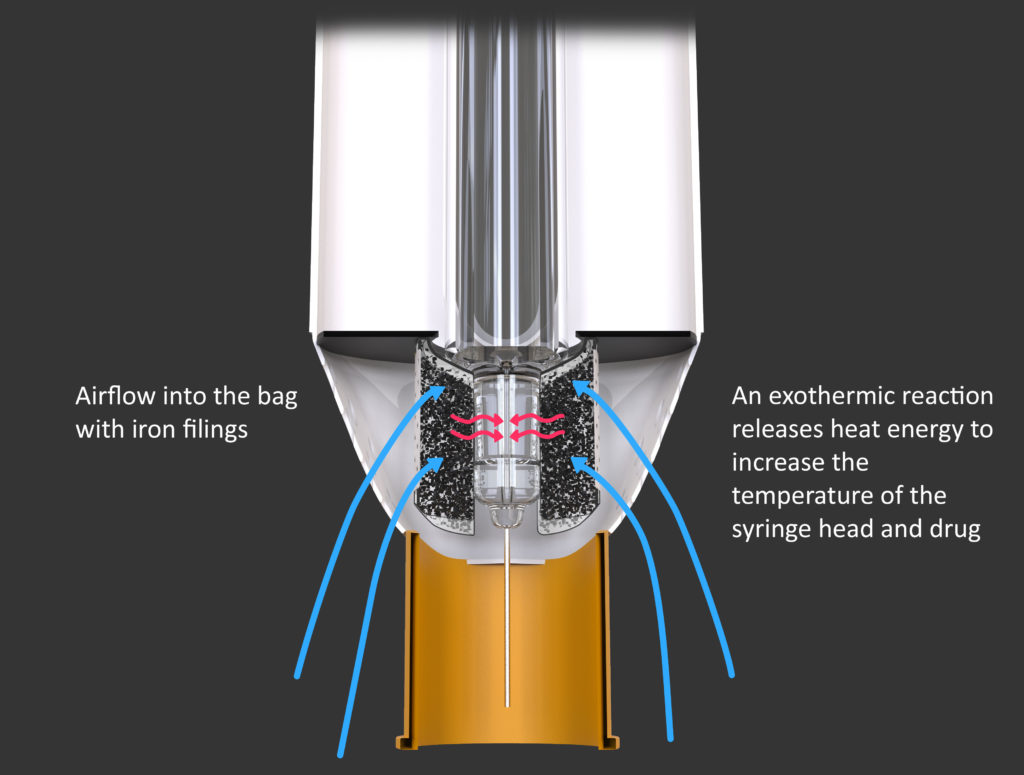
Figure 3: Harnessing the energy from the oxidation of iron to raise the temperature of the drug in an autoinjector. Blue arrows indicate airflow, and red arrows indicate heat transfer.
To ensure this warming process is initiated just before use, the oxidation reaction could be activated when the user removes the device and needle cap (Figure 4). In this example implementation, two bags of iron filings are elasticated together, and are prevented from oxidizing by an air-impermeable film adhered to the bags and the device casework. When the user removes the device cap, the mechanical design causes three things to happen automatically:
- The needle cap is removed;
- the bags of filings are pulled inwards to make contact with the syringe;
- and the air-impermeable film peels off, thereby allowing air to flow through to the iron filings.
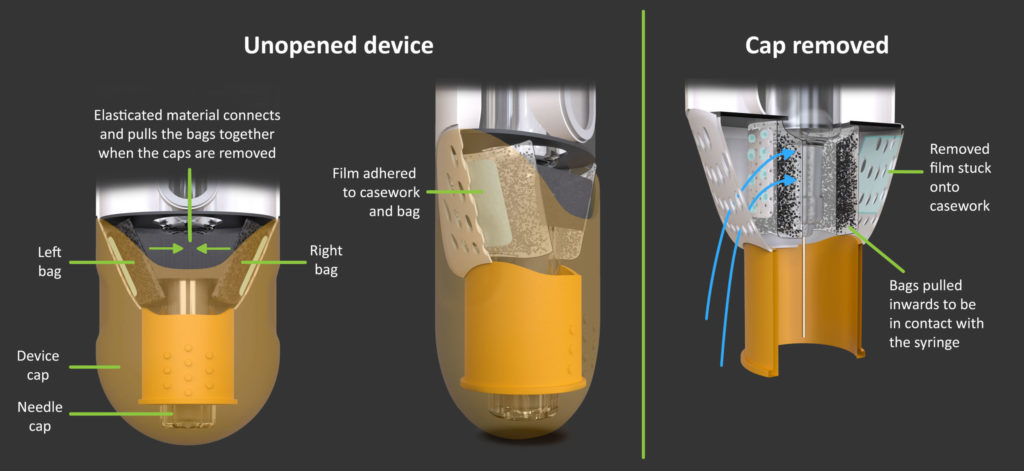
Figure 4: Cap removal activates the warming process by creating an air path to the iron filings.
The user steps of this autoinjector concept are mainly unchanged from traditional devices. The most evident change for the user would be the use of a thermochromic label (changing from grey to orange in Figure 5) to show the user when the drug has reached the optimal temperature for injection.
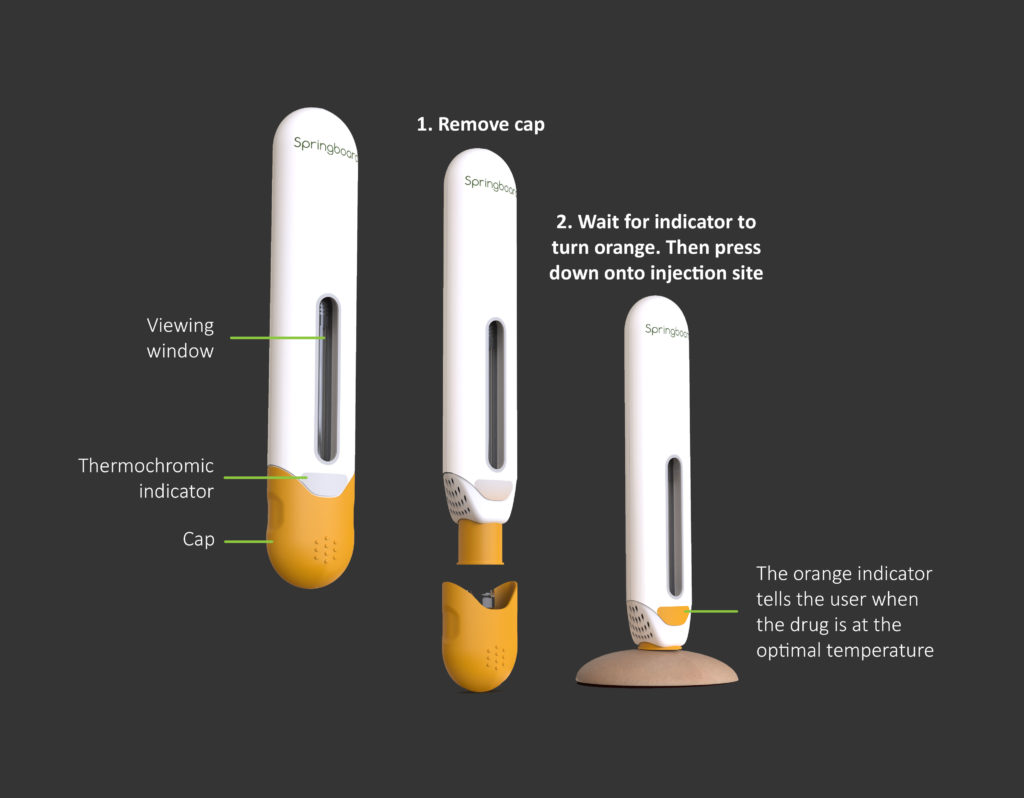
Figure 5: User steps, including a thermochromic indicator that changes from grey to orange.
How can we control the temperature?
It is important to limit the temperature reached by the drug, both for patient comfort and to maintain the chemical integrity of the drug. Our concept makes use of a feedback loop governed automatically by physical fluid properties to limit the temperature to body temperature, i.e. approximately 37 °C.
Initially, the drug is relatively cold and therefore viscous, which makes it flow relatively slowly through the syringe. This means it spends a relatively long time around the bags of iron filings, and so warms up (Figure 6, left). Conversely, once the drug has warmed up, it is less viscous and therefore flows more quickly through the syringe, thereby spending less time around the heating mechanism and absorbing less heat (Figure 6, right).
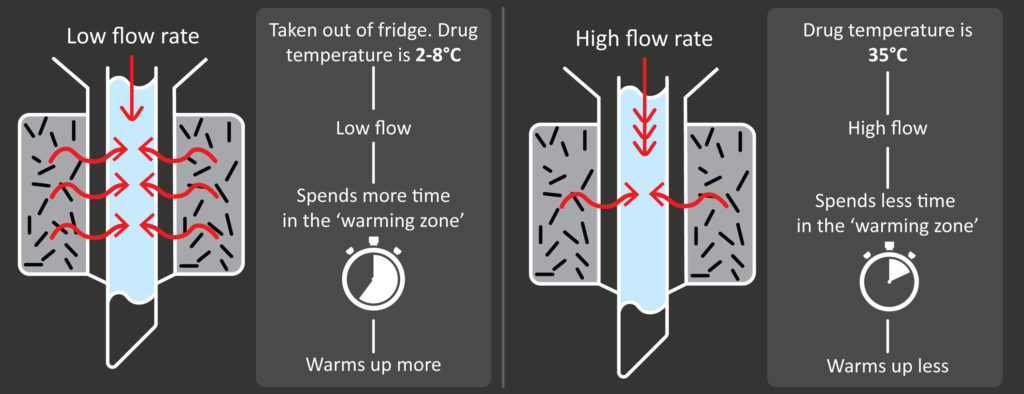
Figure 6: Stages of the physical feedback loop that prevents over-warming of the drug.
In this way, the temperature of the drug is controlled (Figure 7) without the need for sensing or actuating electronics, which would complicate device development and raise the cost of the product.
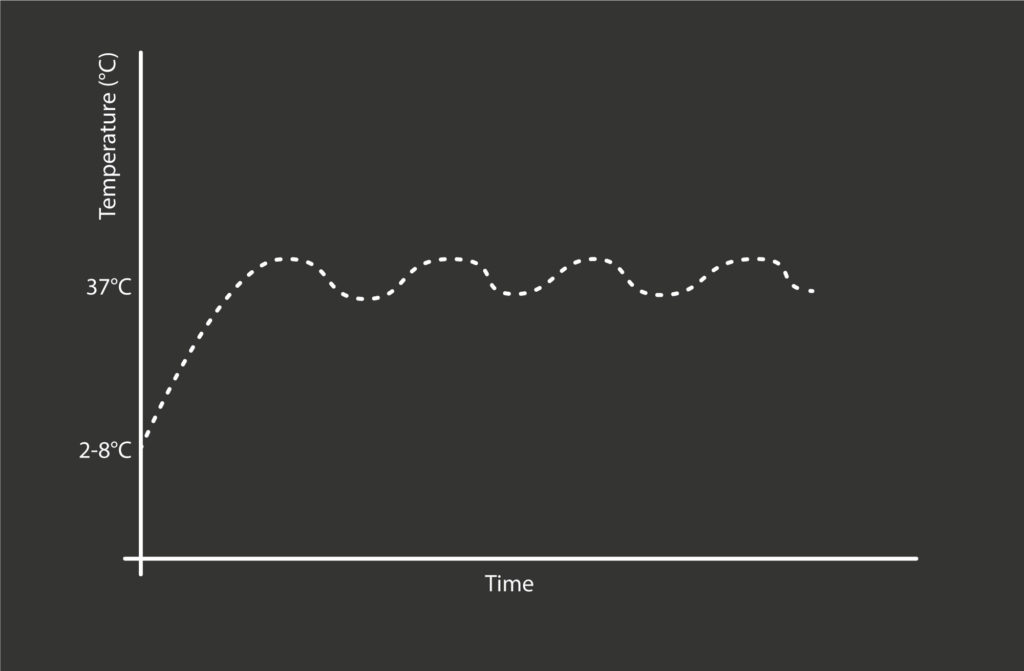
Figure 7: Illustrative graph of temperature of the drug versus time, showing the temperature-limiting effect of the physical feedback loop described in the text.
To achieve a particular warming profile, the specific geometry of the syringe, the amounts and distributions of iron filings, and several other parameters would be optimised by a combination of theoretical modelling, experimentation and knowledge of manufacturability constraints.
Conclusions
This article illustrated just one example of strategic technical innovation. It involved first mapping a user journey to prioritise opportunities for improvement; then creative technical thinking to generate an innovative concept that addresses a user need; and finally realising an optimised design from that concept by using a range of physical sciences and engineering.
Springboard offers this combination of skills, together with extensive industry knowledge of medical devices. If you would like to know more about this concept, or more generally how Springboard an help with your product development process — including concept visualisation, growing our clients’ intellectual property portfolios, and designing parts for manufacture — do not hesitate to get in touch.
References
[1] Business Wire – Autoinjectors market. Accessed October 2021.
[2] Avonex Pen Instructions for Use. Accessed October 2021.
[3] MS Society – Beta interferons. Accessed October 2021.
— Leanne Yip and Tom Oakley


